
MCQs
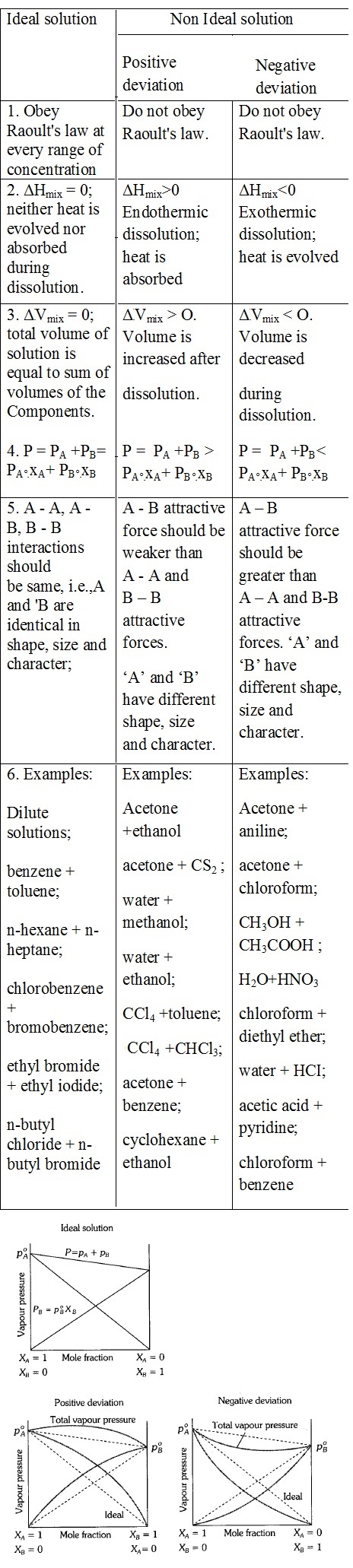
1. An ideal solution obeys Raoult's law:
A. Only at low concentration
B. Only at high concentration
C. At every range of concentration
D. At zero pressure
2. In an ideal solution, the enthalpy of mixing (ΔVmixing) is:
A. Positive
B. Negative
C. Zero
D. Infinite
3. Positive deviation from Raoult's law occurs when:
A. A—B interactions are stronger than A—A and B—B
B. A—B interactions are weaker than A—A and B—B
C. All intermolecular forces are equal
D. Molecules are identical
4. In negative deviation from Raoult's law, the enthalpy of mixing (ΔHmixing) is:
A. Positive
B. Negative
C. Zero
D. Infinite
5. In an ideal solution, the volume change on mixing (ΔVmixing) is:
A. Positive
B. Negative
C. Zero
D. Infinite
6. In positive deviation from Raoult's law, the volume change on mixing is:
A. ΔVmixing = 0
B. ΔVmixing > 0
C. ΔVmixing < 0
D. Infinite
7. Which of the following is an example of an ideal solution?
A. Acetone + ethanol
B. Benzene + toluene
C. Acetone + chloroform
D. Water + HCl
8. Which pair shows positive deviation from Raoult's law?
A. Acetone + ethanol
B. Acetone + chloroform
C. Water + nitric acid
D. Acetone + aniline
9. Which pair shows negative deviation from Raoult's law?
A. Cyclohexane + ethanol
B. Acetone + CS2
C. Acetone + chloroform
D. Water + ethanol